-
Consumer Goods
- Overview
- Industries
- Services
Services
Access a full range of integrated solutions to support your business growth.
- Insights
Insights
Stay updated with our latest insights on Asia and beyond.
-
Healthcare
- Overview
- Industries
- Services
- Insights
Insights
Stay updated with our latest insights on Asia and beyond.
-
Performance Materials
- Overview
- Industries
- Services
- Insights
Insights
Stay updated with our latest insights on Asia and beyond.
-
Technology
- Overview
- Industries
- Services
- Insights
Insights
Stay updated with our latest insights on Asia and beyond.
- Our products
Our products
Search our product database.
-
Services
- Overview
- Sourcing
Sourcing
Accessing a global sourcing network.
- Market insights
Market insights
Generating ideas for growth.
- Marketing and sales
Marketing and sales
Opening up new revenue opportunities.
- Distribution and logistics
Distribution and logistics
Delivering what you need, when you need it, where you need it.
- After-sales services
After-sales services
Servicing throughout the entire lifespan of your product.
- Digital & IT Backbone
DKSH CSSC
Learn about our global hub for Digital & IT services.
- Insights
- Home
- Insights
- Our expertise
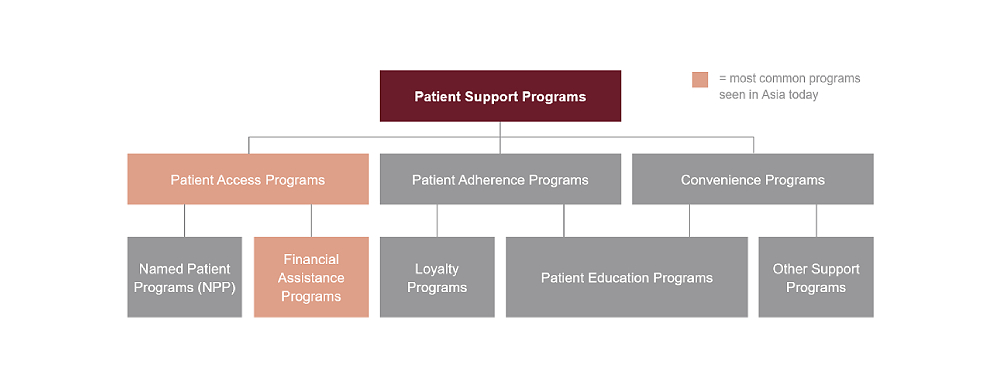
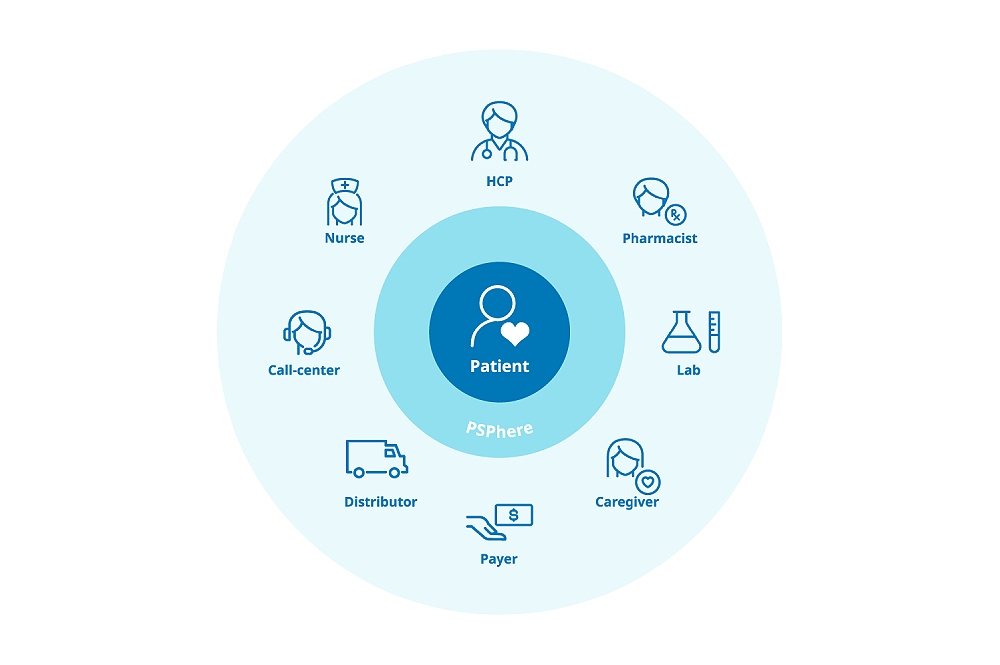
- Four Reasons Why Ethics and Compliance Are Important for Patient Support Programs
- Home
- Insights
- Our expertise
- Four Reasons Why Ethics and Compliance Are Important for Patient Support Programs